Which of the Following Is a Simple Definition of Reduction
From Simple English Wikipedia the free encyclopedia Reduction is a chemical reaction that involves the gaining of electrons by one of the atoms involved in the reaction between two chemicals. Photon and quantum are two very significant principles talked about in modern physics.

Oxidation And Reduction In Organic Chemistry Master Organic Chemistry
These principles are broadly used in quantum chemistry quantum physics electromagnetic theory particle physics optics etc.
. It challenges all standards and makes continuous efforts to better them. Ozonolysis with O 3. 1 on a question Which of the following is a simple definition of reduction.
The large data set D is partitioned into mutually disjoint sets called strata. Hydrogenation is addition of Hydrogen to alkene and alkyne and therefore it is a reduction reaction. The donationloss of electrons.
An increase in the oxidation state exhibited by the substance. Oxygen is usually -2. Thus it can be understood that redox titrations involve a transfer of electrons between the given analyte and the titrant.
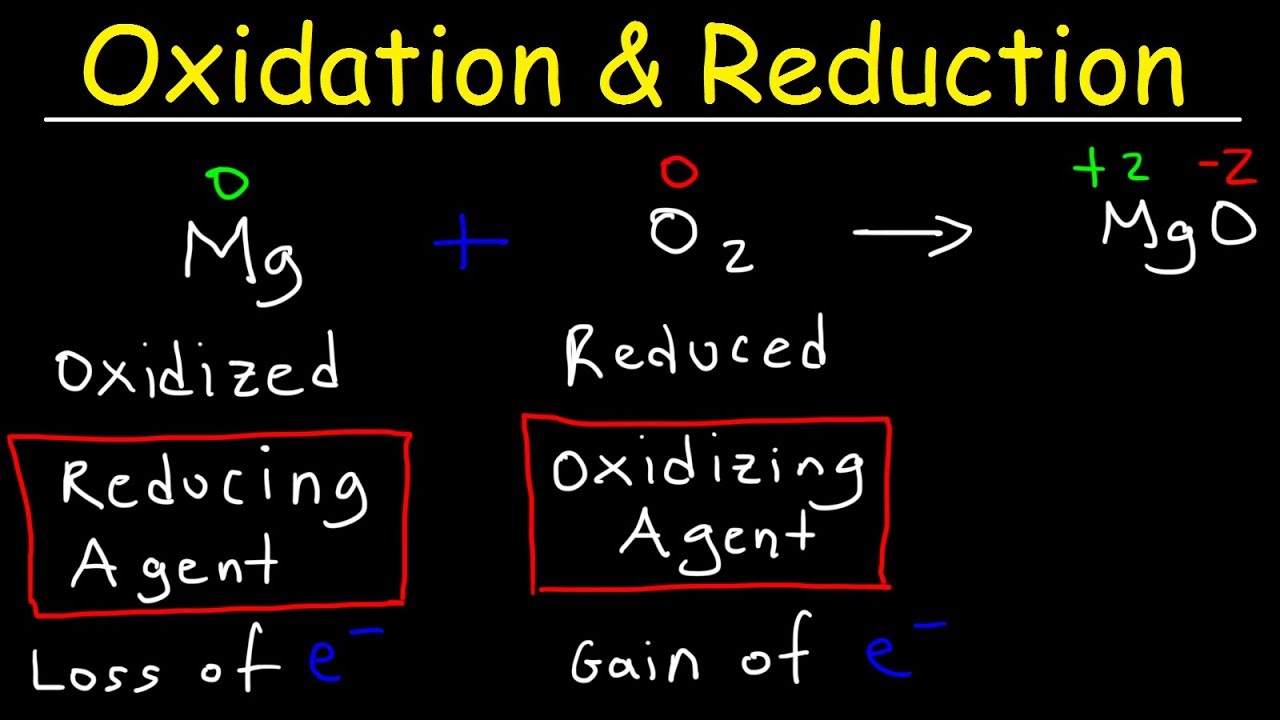
Approach Cost reduction is a continuous process and recognizes no condition as permanent. Oxidation is Removal of electron removal of hydrogen addition of oxygen and increase in oxidation state. Which identifies an oxidation-reduction reaction.
Which of the following is a simple definition of reduction. Solve any question of Redox Reactions with-. Which identifies an oxidation-reduction reaction.
For more than 200 years or since the industrial revolution the concentration of carbon dioxide CO 2 in. Reduction may be considered the opposite process of oxidation. The state of being reduced.
The reduced species receives electrons whereas the oxidised species loses them. Fe2O3 3CO 2Fe 3CO. What is ocean acidification.
3 question Which of the following is a simple definition of reduction. The amount by which something is reduced. Which of the following is a simple definition of oxidation.
The loss of electrons B. A reaction in which oxidation numbers change. The act or process of reducing.
Ocean acidification refers to a reduction in the pH of the ocean over an extended period of time caused primarily by uptake of carbon dioxide CO 2 from the atmosphere. The loss of electrons the gain of electrons an agent that oxidizes something an agent that - 6321322 vinodca3334 vinodca3334 23102018. Which of the following is an oxidation-reduction reaction.
Now a simple random sample is taken from each stratum to get stratified data. The term refers to the element that accepts electrons as the oxidation state of the element that gains electrons is lowered. An agent that oxidizes something D.
A reaction in which oxidation numbers change. 3 Spanish reducción from Latin reduction-. Hydrogenation of alkene and alkyne.
Together reduction and oxidation form redox reactions reduction-oxidation redox. Reduction is Gain of electron and addition of hydrogen Removal of oxygen Decrease in oxidation state. The gain of electrons C.
The other half of the reaction involves oxidation in which electrons are lost. This is a redox reaction in which octane C8H18 is oxidized. An example of a redox titration is the treatment of an iodine solution with a reducing agent.
The industrial establishments comprise reduction works saw-mills and flour-mills one of the latter being the largest in Canada. The data reduction can be applied by implementing SRSWOR on these clusters. But the greatest reduction with only A remaining is characteristic of such a heterogeneous assembly as Accipitres Cypselidae Trochilidae Striges and Fregata.
At the factories the peanuts are packaged at atmospheric conditions whereby P₁ 1 atm however the pressure of the air in the atmosphere decreases with altitude as such the. Which rule for assigning oxidation numbers is correct. Oxidation occurs when the oxidation state of a molecule atom or ion is increased.
Oxidation is the loss of electrons during a reaction by a molecule atom or ion. Objectives Cost Reduction is directed to explore the possibilities of improving the targets or standards themselves. The opposite process is called reduction which occurs when there is a gain of electrons or the oxidation state of an atom molecule or ion decreases.
The gain of electrons. According to Boyles law the pressure of a given mass of gas is inversely proportional to it volume at constant temperature. Reduction involves a half-reaction in which a chemical species decreases its oxidation number usually by gaining electrons.
A reduction-oxidation or redox reaction is a type of chemical reaction in which reduction and oxidation occur at the same time. Zn is oxidative cleavage addition of oxygen and is an oxidation reaction. From these clusters a simple random sample of size s could be generated where s.
Watch our video for a quick overview. Quantum is deemed to be a distinct container with energy kept in whereas photon is considered as an elementary particle. Which of the following is a simple definition of oxidation.
Something made by reducing.

Oxidation Reduction Redox Reactions Balancing Redox Reactions Chemistry Net Chemistry Lessons Chemistry Help Teaching Chemistry

Oxidation And Reduction Reactions Basic Introduction Youtube

Difference Between Oxidation And Reduction Redox Reaction Chemistry Basics Chemistry Lessons Chemistry Education
No comments for "Which of the Following Is a Simple Definition of Reduction"
Post a Comment